Highly Cited
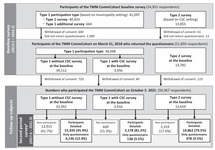
Progress Report of the Tohoku Medical Megabank Community-based Cohort Study: Study Profile of the Repeated Center-based Survey During Second Period in Miyagi Prefecture

A Century of Change: Unraveling the Impact of Socioeconomic/Historical Milestones on Age at Menarche and Other Female Reproductive Factors in Japan
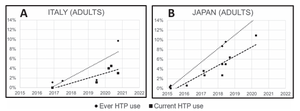
Patterns of Use of Heated Tobacco Products: A Comprehensive Systematic Review
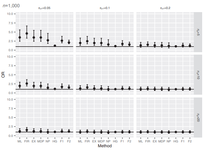
Bias in Odds Ratios From Logistic Regression Methods With Sparse Data Sets
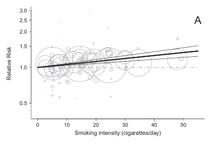
Dose-response Relationships Between Cigarette Smoking and Breast Cancer Risk: A Systematic Review and Meta-analysis
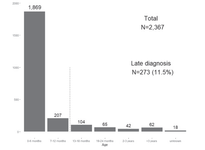
Epidemiology of Developmental Dysplasia of the Hip: Analysis of Japanese National Database
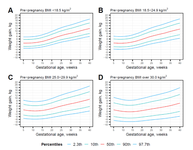
Gestational Weight Gain Growth Charts Adapted to Japanese Pregnancies Using a Bayesian Approach in a Longitudinal Study: The Japan Environment and Children’s Study
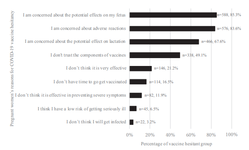
The Prevalence of COVID-19 Vaccination and Vaccine Hesitancy in Pregnant Women: An Internet-based Cross-sectional Study in Japan
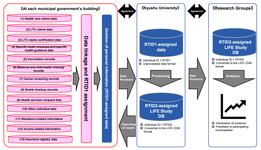
The Longevity Improvement & Fair Evidence (LIFE) Study: Overview of the Study Design and Baseline Participant Profile
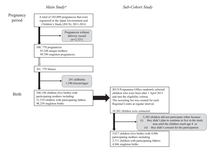
Study Design and Participants’ Profile in the Sub-Cohort Study in the Japan Environment and Children’s Study (JECS)
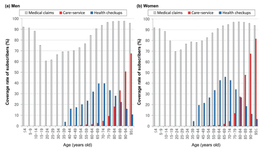
Data Resource Profile of Shizuoka Kokuho Database (SKDB) Using Integrated Health- and Care-insurance Claims and Health Checkups: The Shizuoka Study
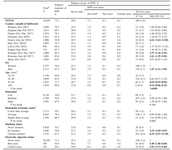
Use and Awareness of Heated Tobacco Products in Europe
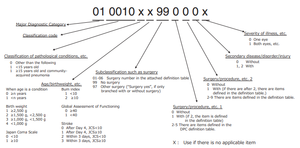
History and Profile of Diagnosis Procedure Combination (DPC): Development of a Real Data Collection System for Acute Inpatient Care in Japan
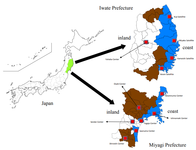
Study Profile of the Tohoku Medical Megabank Community-Based Cohort Study





